""" from atom.api import Atom, Coerced class Demo ( Atom ): # The coercer could be omitted but being explicit helps static type checkers cint = Coerced ( int, coercer = int ) cfloat = Coerced ( float, coercer = float ) cstr = Coerced ( str, coercer = str ) if _name_ = "_main_" : demo = Demo () print ( "CInt Demo" ) demo. # - """Demonstration of the basic use of the Coerced member.
ATOM EXAMPLES FULL
# The full license is in the file LICENSE, distributed with this software.
/atomic-weight-and-atomic-mass-difference-4046144_FINAL_STILL-5940e35000b145ba83fb8e3e40792ba9.png)
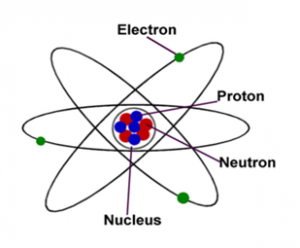
# Distributed under the terms of the Modified BSD License. Any atoms in an ionic/electrovalent bond can get or give up electrons.# - # Copyright (c) 2013-2021, Nucleic Development Team. The attraction of opposite charges is the way they form and maintain the bond. What is atom Meaning of atom medical term. The positive and negative charges continue to attract each other like magnets. Looking for online definition of atom in the Medical Dictionary atom explanation free. When an atom gets an extra electron, it becomes negatively charged like the fluorine ion (F -). Two happy atoms! When an atom gives up an electron, it becomes positive like the sodium ion (Na +). The sodium then has a full second shell and the fluorine (F) also has a full second shell. When they work together, they can both wind up happy! Sodium gives up its extra electron. For example, as a radionuclide decays, it will become a different isotope of the. Everything in the universe (except energy) is made of matter, and, so, everything in the universe is made of atoms. We've also got a fluorine atom that is looking for one. A radioactive atom will attempt to reach stability by ejecting nucleons. So, let’s say we've got a sodium atom that has an extra electron. They can share electrons, making a covalent bond, or they can just borrow them, and make an ionic bond (also called electrovalent bond). There are a couple of ways they can get the electrons. They each have one filled shell with two electrons, but their second shells want to have eight. Each of those elements is looking for a couple of electrons to make a filled shell. For example, when two chlorine atoms are joined by a covalent bond. The main control adopts the ESP32-PICO chip which comes integrated with Wi-Fi. Electronegativity refers to the ability of an atom to attract shared electrons in a. It provides more GPIOs for user customization which is very suitable for embedded smart home devices and in making smart toys. Oxygen (O) and fluorine (F) are two good examples. Atom Lite, which has a size of only 2424mm, is a very compact development board in the M5Stack development kit series. What a coincidence! Many other atoms are interested in gaining a few extra electrons. It is always easier to give away one or two electrons than it is to go out and find six or seven to fill your shells. An atom is the smallest amount of a substance that can take part in a chemical reaction.the news that Einsteins former colleagues Otto Hahn and Fritz. They have two possibilities: they can try to get to eight electrons to fill up their third shell, or they can give up a few electrons and have a filled second shell. You can see that sodium (Na) and magnesium (Mg) have a couple of extra electrons. An atom consists of a central nucleus that is surrounded by one or more negatively. The first shell is filled with 2 electrons, the second is filled with 8 electrons, and the third is filled with 8. An atom is a particle of matter that uniquely defines a chemical element. There is a 2-8-8 rule for these elements.
We should start with the atoms that have atomic numbers between 1 and 18. Those atoms go around looking for other atoms who want to give up an electron. Some atoms are really close to having a full shell. These atoms like to give up their electrons. Some atoms have too many electrons (one or two extra). If you are an atom and you have a shell, you want your shell to be full. The idea behind Happy Atoms is that atomic shells like to be full. We use a concept called "Happy Atoms." We figure that most atoms want to be happy, just like you. You must first learn why atoms bond together.
